Defects from phonons: Atomic transport by concerted motion in simple crystalline metals
E. Fransson
and
P. Erhart
Acta Materialia 196, 770
(2020)
arXiv:1912.01788
doi: 10.1016/j.actamat.2020.06.040
Download PDF
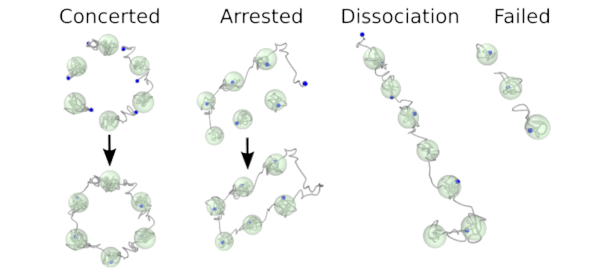
Point defects play a crucial role in crystalline materials as they do not only impact the thermodynamic properties but are also central to kinetic processes. While they are necessary in thermodynamic equilibrium spontaneous defect formation in the bulk is normally considered highly improbable except for temperatures close to the melting point. Here, we demonstrate by means of atomistic simulations that processes involving concerted atomic motion that give rise to defect formation are in fact frequent in body-centered cubic metals even down to about 50% of the melting temperature. It is shown that this behavior is intimately related to the anharmonicity of the lattice vibrations and a flat energy landscape along certain crystallographic directions, a feature that is absent in, e.g., face-centered cubic lattice structures. This insight has implications for our general understanding of these materials and furthermore provides a complementary explanation for the so-called anomalous diffusion in group 4 transition metals.
Associated data
Movie of concerted event Permalink (MP4 movie) Movie of arrested event Permalink (MP4 movie)