Norbornadiene-based photoswitches with exceptional combination of solar spectrum match and long term energy storage
M. Jevric,
A. U. Petersen,
M. Mansø,
S. K. Singh,
Z. Wang,
Ambra,
C. Sumby,
M. Brøndsted Nielsen,
K. Börjesson,
P. Erhart,
and
K. Moth-Poulsen
Chemistry - A European Journal 24, 12767
(2018)
doi: 10.1002/chem.201802932
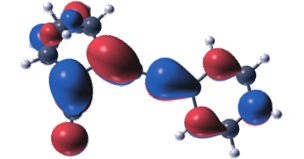
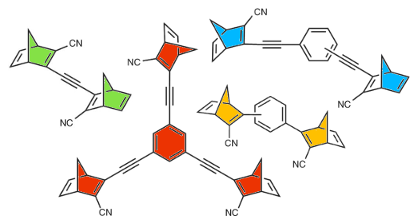
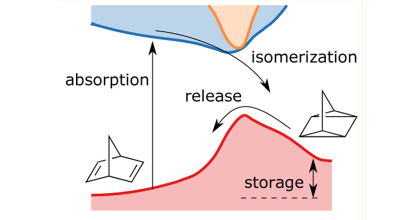
Norbornadiene-quadricyclane (NBD-QC) photo-switches are candidates for applications in solar thermal energy storage. Functionally they rely on an intramolecular [2+2] cycloaddition reaction, which couples the S0 landscape on the NBD side to the S1 landscape on the QC side of the reaction and vice-versa. This commonly results in an unfavourable correlation between the first absorption maximum and the barrier for thermal back-conversion. Here, we demonstrate that this correlation can be counteracted by using steric repulsion to hamper the rotational motion of the side groups along the back-conversion path. It is shown that this modification reduces the correlation between the effective back-conversion barrier and the first absorption maximum and gives rise to a large conversion entropy. The resulting molecules exhibit exceptionally long half-lives for their metastable forms without significantly affecting other properties, most notably solar spectrum match and storage density.