Structural and thermodynamical properties of tungsten oxides from first-principles calculations
C. Linderälv
Master′s Thesis
(2016)
Download PDF

Tungsten as a material has obtained attention because of its prominent role in fusion reactor design, where it is employed in the divertor region of the first wall. In case of an accident, the tungsten tiles may be exposed to atmospheric conditions and high temperatures which would oxidize the material.
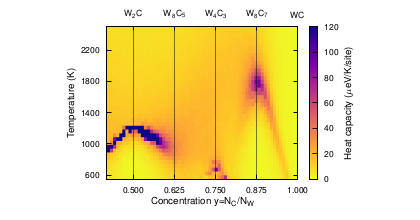
Over the years there has been a lot of studies on tungsten oxides, specially WO3, but not from the perspective of tungsten oxidation. The mechanism of oxidation is unclear and a sound understanding of which oxides are formed is still lacking.
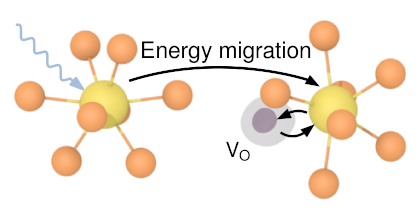
In this work, first principles calculations have been employed to study the relative phase stabilities from a mechanical and thermodynamical perspective. Oxidation has been studied using a point defect approach to the formation and migration energies of oxygen in tungsten and tungsten oxides. Using the same approach, the role of Y substitutional defects has been studied.
Tungsten trioxide is thermodynamically stable with respect to tungsten dioxide but is not as dynamically stable as tungsten dioxide. Oxygen interstitials have a low formation and migration energy in tungsten and several interstitials tend to pair up to form stable clusters, while interstitials in the oxide phases have higher formation energies. In the oxide phases the oxygen vacancy is associated with high formation energies. The difference in phonon frequencies between a pure system and the Y substitutional system is found to be small.
The main oxide, tungsten trioxide, is predicted to be dynamically unstable with imaginary phonon frequencies. Therefore, the active components should be chosen such that the mechanical properties of the formed oxide are enhanced. In this study the Y substitutional is found to have no stabilizing effect on tungsten trioxide, however, the full phonon dispersion for the defect system has not been obtained yet.