Solvent Effects on the Absorption Profile, Kinetic Stability, and Photoisomerization Process of the Norbornadiene-Quadricyclanes System
M. Quant,
A. Hamrin,
A. Lennartson,
P. Erhart,
and
K. Moth-Poulsen
The Journal of Physical Chemistry C 123, 7081
(2019)
doi: 10.1021/acs.jpcc.9b02111
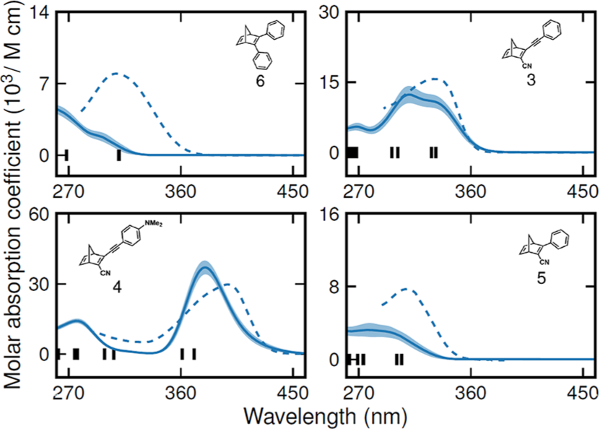
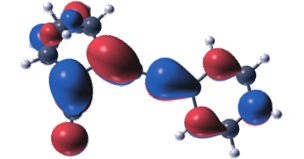
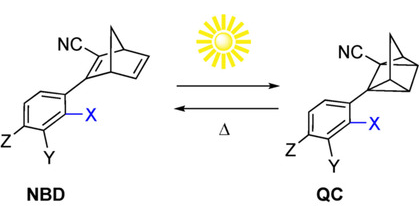
Molecular photoswitches based on the norbornadiene−quadricyclane (NBD-QC) couple can be used to store solar energy and to release the stored energy as heat on demand. In this context, the energy storage time as well as the quantum yield of the energy storing reaction are important parameters. Here, we explore for the first time solvent effects on these processes for a series of four NBD-QC compounds in four different solvents with different polarity (acetonitrile, tetrahydrofuran, toluene, and hexane). We show that the energy storage time of the QC forms can vary by up to a factor of 2 when going from the most to the least polar solvent. Moreover, we show that for norbornadiene derivatives with an asymmetric 1,2 substitution pattern, the quantum yield of conversion is highly solvent dependent, whereas this is not the case for the symmetrically substituted compounds. The spectroscopic observations are further rationalized using classical molecular dynamics (MD) simulations and time-dependent density functional theory (TDDFT) calculations. These demonstrate the importance of vibrational and rotational excitations for obtaining broad-band absorption, which is a prerequisite for capturing a wide range of the solar spectrum.