Thermodynamics of gold nanoparticle growth: A first-principles investigation
H. Shekhar
Master′s Thesis
(2013)
doi: 20.500.12380/194449
Download PDF

Good control over size and shapes of nanoparticles is key for their usage as basic building blocks in various applications. Nanocrystals are of immense interest because of their electronic, optical, and magnetic properties, which are dependent on size and morphology of the nanocrystals. Hence by controlling these parameters it is possible to tune these properties as well. In spite of significant progress in the area of crystallography and materials science the underlying growth mechanism of anisotropic nanocrystals has not been resolved to a satisfactory level. Proper understanding of growth and control mechanism is key for further improvement, which will open the door for applications in new areas.
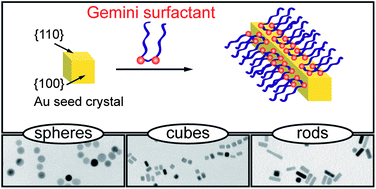
Gold nanorods are examples for anisotropic nanocrystals which is a topic of research for many groups. Bottom-up, e.g., templating, electrochemical and top-down, e.g., photolithography are two methods for the synthesis of metal nanoparticles. One of the most widely used methods for the synthesis of anisotropic gold nanorods is surfactant assisted seed mediated growth from solution. Seed mediated growth is a wet chemical method is a cheap and easy process. While it offers several advantages over electrochemical processes, it suffers from low yield and the unpredictible morphology of the end product in many cases.
The objective of this thesis is to resolve the thermodynamics of the growth of Au nanoparticles and rods using cetyltrimethylammonium bromide as growth agent. To this end the surface energetics of a variety of different Au surfaces with and without surfactant are obtained using electronic structure calculations based on density functional theory. The results are discussed in the context of experimental observations. The growth of nanocrystals is not an equilibrium phenomena so its growth mechanism cannot be explained by pure thermodynamics alone. The present study provides basic thermodynamic parameters, which determine the driving forces and exact knowledge of which is a prerequisite for understanding the non-equilibrium growth process. Thereby present work lays the ground for future studies that should address, for example, entropic and kinetic factors in order to get a comprehensive picture of nanocrystal and nanorod formation.