Revealing the kinetics of interfacial surfactant phase transitions through multiscale simulations and in-situ plasmonic sensing
E. Berger,
N. Khosravian,
F. A. A. Nugroho,
J. Löfgren,
C. Langhammer,
and
P. Erhart
arXiv:2605.01458
doi: 10.48550/arXiv.2605.01458
Download PDF
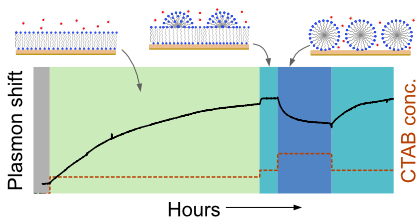
Surfactant self-assembly at solid-liquid interfaces governs interfacial stability, transport, and reactivity across many technologies, yet resolving interfacial surfactant phases and their transition kinetics in situ remains challenging. Here, we establish an atomistically grounded plasmonic framework that quantitatively maps interfacial surfactant phases and phase transitions onto optical signatures. Distinct morphologies differ in packing and hydration, modifying the effective permittivity within the optical near field and producing surfactant phase-specific plasmonic extinction peak shifts. Using cetyltrimethylammonium bromide on silica as a prototypical surfactant-surface system, we combine atomistic simulations, electronic-structure calculations, and continuum electrodynamics to translate molecular morphologies into predicted spectral shifts for literature-reported surface phases. We experimentally confirm the predicted ordering and magnitude of steady-state peak shifts during stepwise concentration changes, and extract transition kinetics from exponential relaxations of the time-resolved peak shift. A key mechanistic signature is reversal of the spectral shift direction upon transition from an impermeable bilayer to a water-accessible, channel-containing phase, consistent with hydration-driven reduction of the local effective permittivity. Because the approach relies on dielectric contrast in the plasmonic near field and works through a dielectric overlayer, it provides a broadly applicable route for real-time identification of interfacial surfactant phases and their kinetics in aqueous conditions.