Competing adsorption of H and CO on Pd-alloy surfaces: Mechanistic insight into the mitigating effect of Cu on CO poisoning
P. Ekborg-Tanner
and
P. Erhart
arXiv:2603.00776
zenodo: 17670908
(associated data)
Download PDF
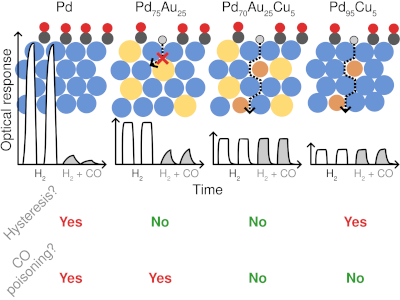
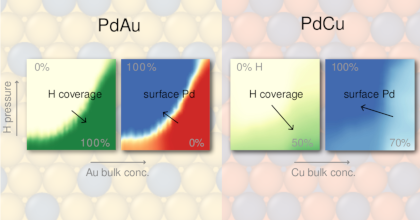
Multi-component alloys offer broad tunability for addressing challenges in materials science, but their vast configurational space makes their surface chemistry highly sensitive to operating conditions, for example through adsorption and segregation. Here, we study Pd-Au-Cu alloy surfaces in H2 and CO environments motivated by their use in H technologies, in particular plasmonic H2 sensing, where alloying can mitigate limitations intrinsic to Pd such as hysteresis and CO poisoning. Modeling multicomponent surfaces with multiple adsorbate species under realistic conditions is challenging. To this end, we establish an accurate and efficient framework that combines machine-learned interatomic potentials trained on density functional theory data to generate training data for cluster expansions with effectively no limitations on training set size. By constructing continuous surface phase diagrams for H-CO coadsorption we find that coadsorption under operating conditions is governed primarily by the H coverage during annealing. Au-rich surfaces, formed under H-poor conditions, suppress both CO and H adsorption, while H-rich conditions yield Pd-rich surfaces that maintain higher H coverages compared to Pd at relevant CO partial pressures, indicating improved CO poisoning resistance. This effect is insensitive to relative amounts of Au and Cu, despite experimental evidence of the mitigating effect of specifically Cu on CO poisoning. Kinetic barriers for dilute alloy surfaces indicate that absorption pathways near Au are highly unfavorable, while Cu leave the energetics unchanged compared to pure Pd. This finding suggests that Cu in the surface region provides viable pathways to shuttle H into the material when Pd-dominated paths are blocked by CO.